A 1938 mandate that required all experimental drugs to be tested on animals before being used in clinical trials on humans has been scrapped.
The new FDA Modernization Act 2.0, which was signed into law in December 2022 allows drug companies to use other methods of testing, such as miniature tissue models and microfluidic chips, which mimic organ functions by using human cells.
It is estimated that around 50 million animals are tested on every year in the United States of America. Whilst this law doesn’t ban animal testing, it is expected it will significantly reduce the number of animals tested on each year.
As well as being inhumane, animal testing can also be ineffective. Many drugs that work in mice aren’t effective, and can even sometimes be toxic in people.
It’s estimated that up to 90 percent of drug candidates that are in the clinical trial stage never make it to market due to being ineffective or dangerous in humans after passing in animal studies.
Paul Locke, scientist and lawyer at Johns Hopkins University who studies alternatives to animal testing said:
“We have many important drugs that have been developed using animal tests. But as we get into some of these more difficult diseases, especially neurological diseases, the animal models just aren’t serving us as well”
Loke went on to say, “We need new ways to really unlock the molecular mechanisms that are causing these diseases, and the alternatives I think hold great promise.”
What are the alternatives?
There are many alternatives to animal testing, with many of those actually outperforming animal testing for reliability for determining safety in humans. Below are just three examples of viable alternatives currently in use, with many more options currently under development.
Organ on a chip

Organs on chips are systems that contain miniature human tissues and fluids inside a microfluidic chip. One example is the liver on a chip by Emulate, which allows researchers to test their treatments for reactions directly on the chip.
Cell cultures
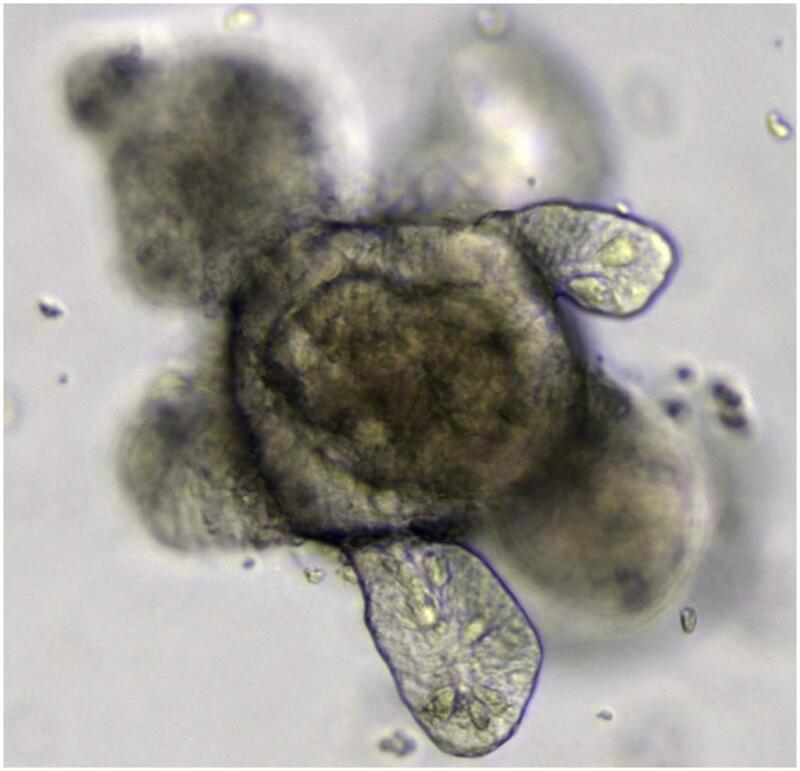
Cell cultures called Organoids, which are hollow 3D clusters of cells that are derived from stem cells and mimic specific tissues. They have shown to be effective in predicting liver and cardiac toxicities. They can be tailored to a patient to test the effect of a drug specifically on cells from their organ.
Computer Models

Computer models refers to software that is designed to emulate Human and Animal bodies and organs to determine both effectiveness and identify risks. One example is the Artificial intelligence system created by CytoReason which is already being used in hospitals.

